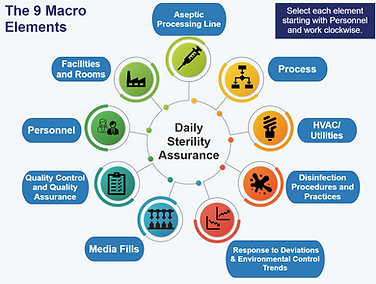
Macro Model Diagram Screenshot
Systems-based macro model illustrating the interdependencies that contribute to daily sterility assurance.
Overview
This short-form web-based training module introduces learners to the 9 macro elements that collectively determine sterility assurance in aseptic pharmaceutical manufacturing.
The Problem
Aseptic processing is inherently complex and multifactorial. The macro elements model includes interdependencies across:
-
Personnel
-
Facilities & Rooms
-
Aseptic Processing Line
-
The Process
-
HVAC & Utilities
-
Disinfection Practices
-
Deviations & Environmental Trends
-
Media Fills
-
Quality Control & Assurance
Learners needed:
-
A conceptual framework before diving into dense guidance documents
-
Clear understanding of contamination risk pathways
-
Awareness of system interdependencies
-
Reinforcement of GMP mindset and quality oversight
Traditional slide-based instruction often overwhelms learners with detail before establishing structure.
Design Approach
This module was designed as a structured conceptual introduction rather
than a regulatory deep dive.
Key Design Strategies
1. Macro Model First, Micro Later
The course establishes a systems-based model of daily sterility assurance
before exploring individual elements. This scaffolding approach reduces
cognitive overload and improves knowledge transfer.
2. Progressive Disclosure & Navigation Control
Slides require full engagement before progression (as noted in the navigation guidance, ensuring learners process each concept intentionally.
3. Visual Reinforcement of Interdependencies
The recurring Macro Model diagram visually reinforces the interrelationship between elements - while reinforcing the 9 elements.
4. Risk-Based Framing
Content consistently ties operational elements back to contamination risk and patient safety impact, reinforcing GMP accountability.
5. Structured Knowledge Checks
A 4-question graded assessment (75% passing threshold) reinforces conceptual understanding rather than rote recall.
Instructional Design Decisions
-
Balanced regulatory language with simplified conceptual framing
-
Used scenario-driven phrasing (e.g., contamination hazards, deviation impacts)
-
Reinforced contamination risk pathways across multiple slides
-
Designed consistent feedback layers for correct/incorrect responses
-
Structured module length (~15 minutes) for microlearning-style deployment
Accessibility Considerations
-
Structured slide navigation
-
Clear heading hierarchy
-
Layered feedback for knowledge checks
-
Designed in Articulate Storyline (HTML5 output)
Outcome
This module became the foundational primer for subsequent GMP
and aseptic processing training initiatives.
Post-launch, the client reported above-average employee satisfaction
and accelerated concept adoption.


Branching Interaction Screenshot
Branching interaction guiding learners through contamination risk pathways and decision impact.
Heavy Content Streamlined
Streamlined compliance-heavy content into structured, accessible language while preserving regulatory intent.
Heavy Content Streamlined
Streamlined compliance-heavy content into structured, accessible language while preserving regulatory intent.

Knowledge Check Screenshot
Knowledge check reinforcing critical takeaways required for progression into the next course.
